 When hydrogen peroxide is in its pure form it is usually seen as a clear liquid with a slight pale blue colouration. Its chemical formula is written as H 2 O 2. It has physical properties that are very similar to water, with the exception that it. H 2 O 2 (hydrogen peroxide) is a colourless liquid that is similar to water in several ways. Anhydrous hydrogen peroxide is a colourless, syrupy liquid that decomposes into oxygen and water very easily. The precipitation reaction of ( Remark: Graphics omitted.) is taken advantage of in a new volumetric analysis suited for determination of titanium in quantities of the order of 10 mg. Hydrogen peroxide is a chemical compound containing molecules of hydrogen and water. Hydrogen peroxide is a hydrogen-oxygen chemical compound. A solution containing 2+ remains unchanged but a solution containing + gives a yellow precipitate TiO(OH)(OOH), and one containing 0 or 2− gives a pale yellow compound ( Remark: Graphics omitted.) at room temperature or more quickly when heated on a water-bath. All the tentative formulae of these species are based on the composition of isolated salts, the absorption spectra of the solutions etc. At higher pH values (10 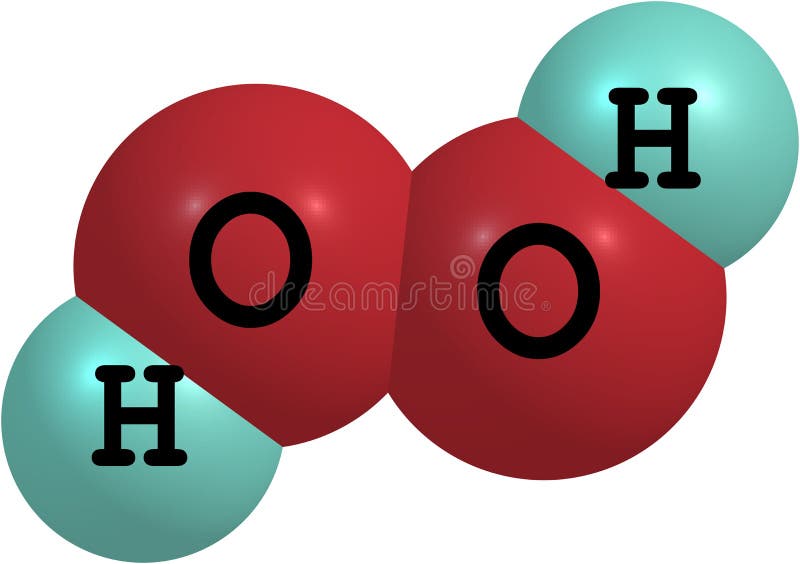 HYDROGEN PEROXIDE (>60 SOLUTION IN WATER), ICSC: 0164. Do not stand over the reaction – steam and oxygen are quickly produced. NO contact with incompatible materials: See Chemical Dangers.  It is used as an oxidizer, bleaching agent and disinfectant. H2 O2 is simple peroxide (a compound with an oxygen-oxygen single bond). In its pure form, the liquid with an odor is slightly more viscous than water. Hydrogen peroxide is a chemical compound with the formula H2O2. The possible presence of a brown color in the foam is evidence of iodine in the reaction vessel.Ĭare should be taken handling 30% hydrogen peroxide – it is severely corrosive to the skin, eyes and respiratory tract. The viscosity value of hydrogen peroxide is higher than that of water. Since this is an exothermic reaction, steam may be seen emerging from the cylinder. The generated oxygen interacts with the soap creating a foam “snake†which quickly fills the cylinder and spills onto the large serving tray. Quickly pour the 2 M KI solution into the 500 mL graduated cylinder containing the hydrogen peroxide plus soap and stand back. Measure 5 mL of 2 M KI solution in 10 mL graduated cylinder. Add 5 mL dishwashing liquid and 10 drops food coloring to the graduated cylinder and shake to mix contents, but not too vigorously to avoid foaming. Measure 20 mL of 30% hydrogen peroxide into the graduated cylinder.Large serving tray and rubber gloves to assist in cleanup.  Dishwashing liquid – approximately 5 mL.Potassium iodide 2 M – Prepare stock solution by mixing 33.2 g KI in distilled water and dilute to 100 mL.20 mL 30% hydrogen peroxide, available from chemical supply establishments.The decomposition of hydrogen peroxide in the presence of iodide ion occurs in two steps: The rapid evolution of oxygen gas is produced by the following reaction: Procedure presents an example of a catalyzed reaction. This is the classic “Elephant Toothpaste†reaction. Two solutions are mixed resulting in an eruption of foam resembling a huge stream of toothpaste.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
